Composition of the gas phase over SiO2/C for a SiO 2 /C mol ratio 1: 3... | Download Scientific Diagram

balancing and writing 3parts 1 .doc - Name: Seat Balance the following equations: 1. 2. P4 O2 P4O10 Zn HCl ZnCl2 H2 3. SiO2 C SiC CO 4. MgO | Course Hero

Titanium, which is used to make airplane engines and frames can be obtained from titanium tetrachloride, which in turn is obtained from titanium oxide by the following process: 3TiO2(s) + 4C(s) +

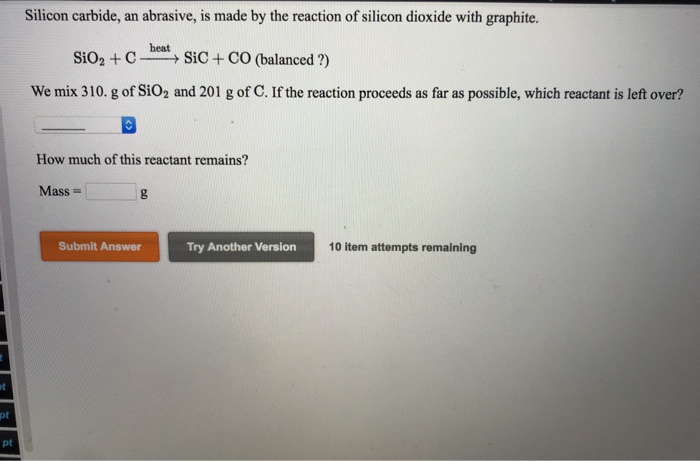
SOLVED: Silicon carbide; an abrasive; is made by the reaction of silicon dioxide with graphite. heat SiO2 + C SiC + CO (balanced ?) We mix 290. g of SiOz and 217

SOLVED: Silicon carbide; an abrasive; is made by the reaction of silicon dioxide with graphite. heat SiO2 + C SiC + CO (balanced ?) We mix 290. g of SiOz and 217
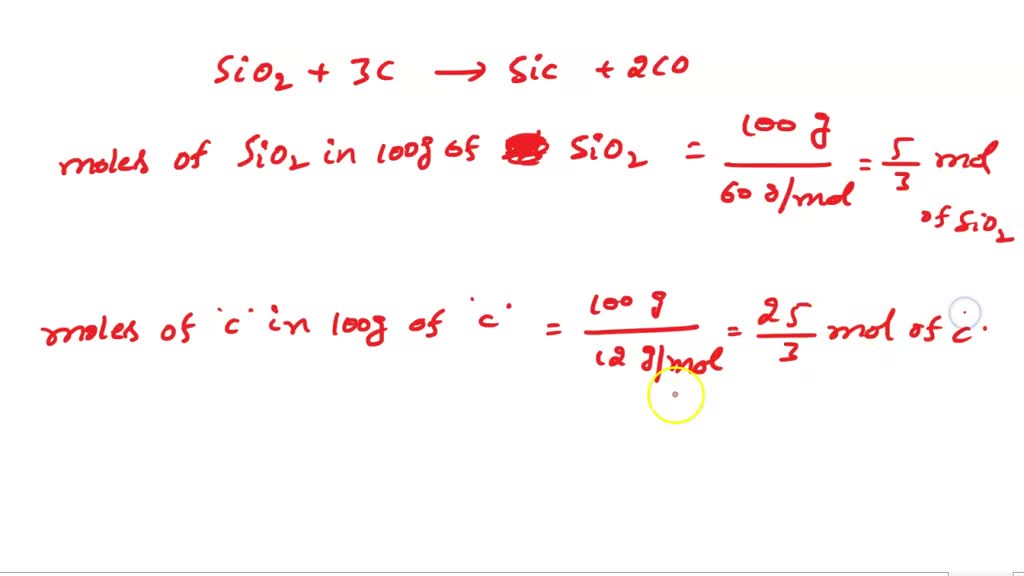
SOLVED: Question 32 5 pts Silicon carbide an abrasive; is made by the reaction of silicon dioxide with graphite SiOz +3C SiC 2CO If 100 g of SiOz and 100 g of

Stoichiometry Notes Chemistry Mrs. Stoops. Stoichiometry Notes Converts grams of one substance into a different substance. Uses: Balanced chemical equations. - ppt download
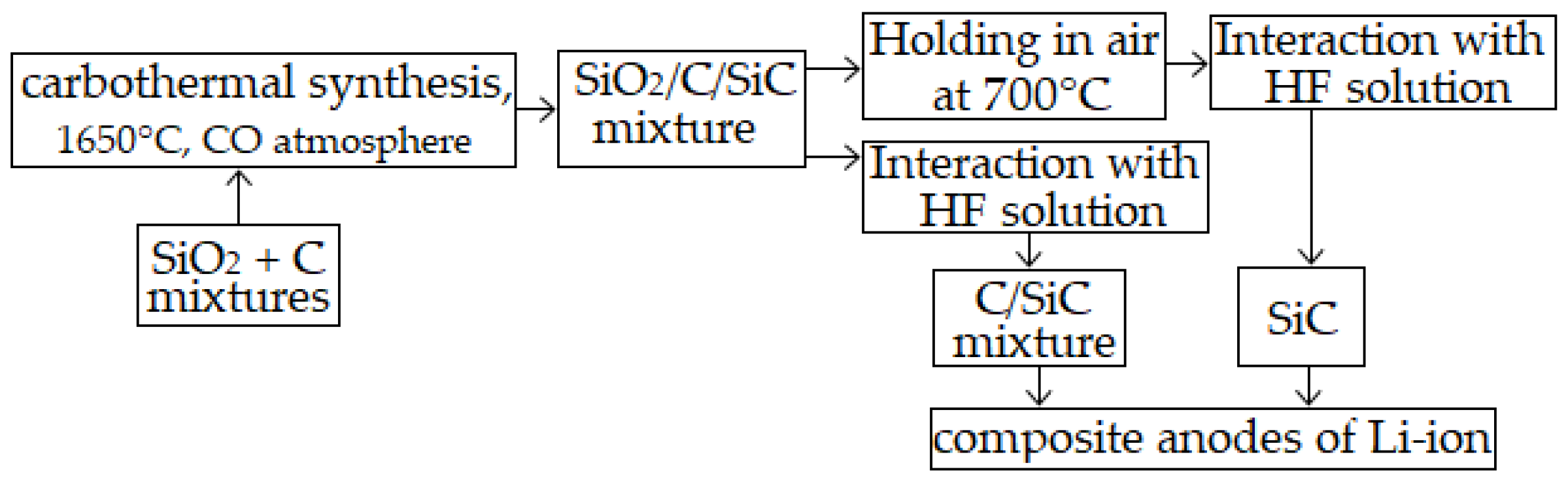
Applied Sciences | Free Full-Text | Synthesis of C/SiC Mixtures for Composite Anodes of Lithium-Ion Power Sources

From the following data at 25C, Calculate the standard enthalpy of formation of FeO(s) and of Fe2O3 (s) .Reaction ΔrH (KJ/mole) (1) Fe2O3 (s) + 3C (graphite) → 2Fe (s) + CO (

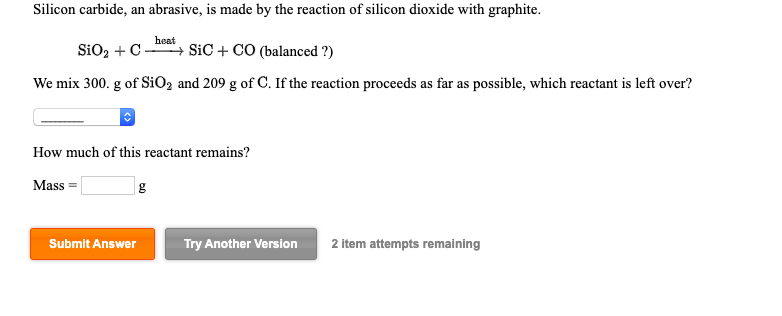
SOLVED: Silicon carbide, an abrasive, is made by the reaction of silicon dioxide with graphite. heat SiOz + C SiC CO (balanced 2) We mix 270. g of SiOz and 207 g

Synthesis of Highly Nanoporous β-Silicon Carbide from Corn Stover and Sandstone | ACS Sustainable Chemistry & Engineering

From the thermo chemical reactions, C(graphite) + 1/2O2→ CO;Δ H = - 110.5KJ CO + 1/2O2→ CO2;Δ H = - 283.2KJ. Δ H for the reaction, C(graphite) + O2→ CO2 is: